
While diluting a solution of salt in water, a student by mistake added acetone (boiling point 56☌). This process is used to obtain wines from the sugarcane juice. What do you understand by the term distillation? Give its one application.ĭistillation is a process of boiling a liquid and then condensing the vapour obtained into a liquid. The only difference is that the dispersed phase in fog is liquid and in smoke it is a solid. In what way are they different? īoth fog and smoke have gas as the dispersion medium. True solutions are vinegar and glucose solution. Pond water, fog, aluminium paint, vinegar and glucose solution. Identify colloids and true solutions from the following: 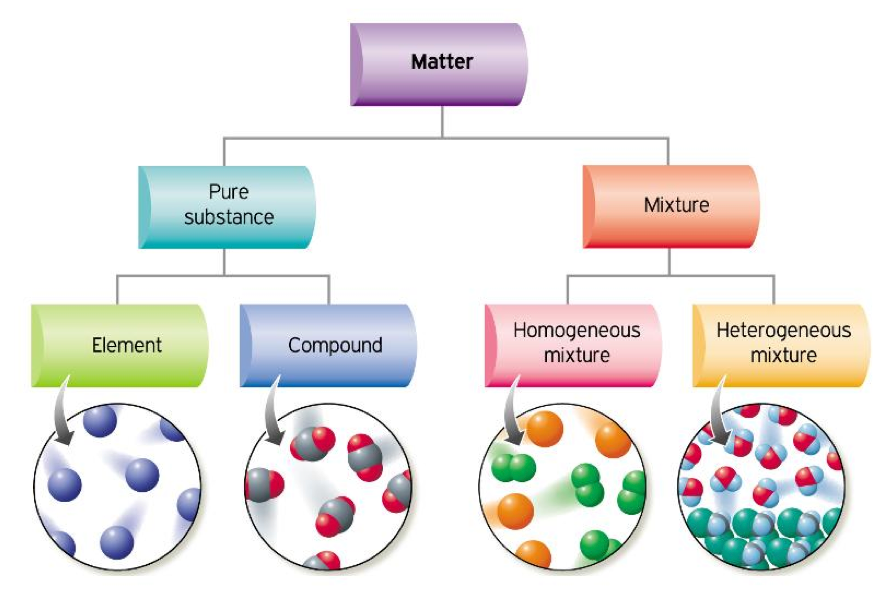
Also molecular interaction in a suspension is not strong enough to keep the particles suspended and hence they settle down. Particle size in a suspension is larger than those in a colloidal solution. They cannot be distinguished even under a microscope.Įxplain why particles of a colloidal solution do not settle down when left undisturbed, while in the case of a suspension they do. The solute and solvent particles are very small. Why is it not possible to distinguish particles of a solute from the solvent in solution?Ī true solution is homogeneous in nature. Sea water also contains mud, decayed plant, etc., other than salts and water, so it is heterogeneous also. Therefore, sea water is considered homogeneous. Sea water is a mixture of salts and water which cannot be separated except by evaporation. ‘Sea water can be classified as homogeneous as well as heterogeneous mixture.’ Comment. Is Matter Around Us Pure Class 9 Extra Questions Short Answer Questions-I Identify the elements from the following substances: sulphur, brine, hydrochloric acid, water, neon, paper, sugar. Unknown substance ‘A’ should be a compound because elements and mixture do not decompose. What is ‘A’-an element, a compound or a mixture? Gallium and mercury exist in liquid state at room temperature.Īn unknown substance ‘A’ on thermal decomposition produces ‘B’ and ‘C’. Name two elements which exist in liquid state at room temperature. Melting of iron metal, bending of iron rod and drawing a wire of iron metal are physical changes because there is no change in the chemical composition of iron, only its form is changing. Melting of iron metal, rusting of iron, bending of iron rod, drawing a wire of iron metal. Which of the following are physical changes? Water is known as universal solvent because it has the ability to dissolve wide variety of substances. On evaporation, these contaminate the solid. some impurities may remain dissolved in the solution even after filtration.some solids decompose or get charred on heating to dryness.Why is crystallisation technique considered better than simple evaporation to purify solids?Ĭrystallisation is considered better than simple evaporation because It is a process that separates a pure solid in the form of its crystals from a solution. So, the mixture of liquids ‘A’ and ‘B’ can be separated by fractional distillation. We can separate two miscible liquids by fractional distillation, if both the liquids have difference in their boiling points. Which separation technique will you apply to separate the mixture of ‘A’ and ‘B’ if the difference in their boiling points is 27☌? Two liquids ‘A’ and ‘B’ are miscible with each other at room temperature. In what respect does a true solution differ from a colloidal solution?Ī true solution is homogeneous whereas a colloidal solution is heterogeneous. The two components of a colloidal solution are dispersed phase and dispersing medium. What are the two components of a colloidal solution? It is defined as the mass in grams of the solute present in one hundred grams of a solution. Iodine is the solute and alcohol is the solvent. Identify solute and solvent in ‘tincture of iodine’. It is a pure substance because chemical composition of sugar crystals is same irrespective of its source.īased on which factor a solution is said to be diluted, concentrated or saturated?Ī solution is said to be diluted, concentrated or saturated on the basis of the amount of solute dissolved in the solution. Will it be a pure substance or a mixture? Give reasons for the same. Sucrose (sugar) crystals obtained from sugarcane and beetroot are mixed together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
